Linus Pauling Was Born #OTD In 1901. He’s Best Known For His Work On Chemical Bonding And The Electronegativity

Linus Pauling was born #OTD in 1901. He’s best known for his work on chemical bonding and the electronegativity scale that bears his name, for which he won a Nobel Prize. Just don’t mention the vitamin C years 😉 https://ift.tt/3uFpNkF https://ift.tt/3r4nm98
More Posts from Amateurchemstudent and Others


Follow @productive-tips for more tips and content like this posted daily! Handpicked and curated with love :)
Covalent and Dative Bonds
Covalent and dative (sometimes called co-ordinate) bonds occur between two or more non-metals, e.g. carbon dioxide, water, methane and even diamond. But what actually are they?
A covalent bond is a chemical bond that involves the sharing of electron pairs between atoms. They are found in molecular elements or compounds such as chlorine or sulfur, but also in macromolecular elements and compounds like SiO2 and graphite. Covalent bonds are also found in molecular ions such as NH4+ and HCO3-.
Single covalent bonds have just one shared pair of electrons. Regularly, each atom provides one unpaired electron (the amount of unpaired electrons is usually equal to the number of covalent bonds which can be made) in the bond. Double covalent bonds have two shared pairs of electrons, represented by a double line between atoms, for example, O=C=O (CO2). Triple covalent bonds can also occur such as those in N ≡ N.
Dot and cross diagrams represent the arrangement of electrons in covalently bonded molecules. A shared pair of electrons is represented by a dot and a cross to show that the electrons come from different atoms.
Unpaired electrons are used to form covalent bonds as previously mentioned. The unpaired electrons in orbitals of one atom can be shared with another unpaired electron in an orbital but sometimes atoms can promote electrons into unoccupied orbitals in the same energy level to form more bonds. This does not always occur, however, meaning different compounds can be formed - PCl3 and PCl4 are examples of this.

An example where promotion is used is in sulfur hexafluoride (SF6). The regular configuration of sulfur atoms is 1s2 2s2 2p6 3s2 3p4. It promotes, as shown in the diagram (see excited state), two electrons: one from the 3s electrons to the 3d orbital and one from the 3p to the 3d. Therefore there are 6 unpaired electrons for fluorine atoms to join. It has an octahedral structure.

An atom which has a lone pair (a pair of electrons uninvolved in bonding) of electrons can form a coordinate bond with the empty orbital of another atom. It essentially donates an electron into this orbital which when formed, acts the same as a normal covalent bond. A coordinate bond therefore contains a shared pair of electrons that have come from one atom.

When ammonia reacts with a H+ ion, a coordinate bond is formed between the lone pair on the ammonia molecule and the empty 1s sub-shell in the H+ ion. An arrow represents the dative covalent bond (coordinate bond). Charges on the final ion must be showed.
Summary
A covalent bond is a chemical bond that involves the sharing of electron pairs between atoms. They are found in molecular elements or compounds as well as in macromolecular elements and compounds. Also found in molecular ions.
Single covalent bonds have just one shared pair of electrons. Double covalent bonds have two shared pairs of electrons, represented by a double line between atoms. Triple covalent bonds can also occur.
Dot and cross diagrams represent the arrangement of electrons in covalently bonded molecules. A shared pair of electrons is represented by a dot and a cross to show that the electrons come from different atoms.
Unpaired electrons are used to form covalent bonds - they can be shared with another unpaired electron in an orbital but sometimes atoms can promote electrons into unoccupied orbitals in the same energy level to form more bonds. This does not always occur, however, meaning different compounds can be formed.
An example where promotion is used is in sulfur hexafluoride (SF6).
An atom which has a lone pair (a pair of electrons uninvolved in bonding) of electrons can form a coordinate bond with the empty orbital of another atom.
It donates an electron into this orbital which when formed, acts the same as a normal covalent bond. A coordinate bond therefore contains a shared pair of electrons that have come from one atom.
When ammonia reacts with a H+ ion, a coordinate bond is formed between the lone pair on the ammonia molecule and the empty 1s sub-shell in the H+ ion. An arrow represents the dative covalent bond (coordinate bond). Charges on the final ion must be showed.


Around a year ago, scientists determined the structure of the SARS-CoV-2 spike protein. Here’s a look at how it was done and how it helped the fight against #COVID19 in the latest edition of #ChemVsCOVID with the Royal Society of Chemistry: https://ift.tt/3pZiZe9 https://ift.tt/3002NPh
Covalent Bonds: Sharing Is Caring!
Welcome to my second out of three posts on bonding - ionic, covalent and metallic. This post also covers the coordinate/ dative bond which I can’t remember if I’ve covered before. Only one more of this series left! Find the others here.
Covalent bonding involves one or more shared pairs of electrons between two atoms. These can be found in simple molecular elements and compounds like CO2 , macromolecular structures like diamond and molecular ions such as ammonium. Covalent bonds mostly occur between non-metals but sometimes metals can form covalent bonds.
Single covalent bonds share just one pair of electrons. Double covalent bonds share two. Triple covalent bonds share three.
Each atom usually provides one electron – unpaired in the orbital – in the bond. The number of unpaired electrons in an atom usually shows how many bonds it can make but sometimes atoms promote electrons to fit in more. Covalent bonds are represented with lines between the atoms – double and triple bonds represented with two and three lines respectively.
Dot and cross diagrams show the arrangement of electrons in covalent bonds. They use dots and crosses to demonstrate that the electrons come from different places and often only the outer shell is shown.

The simple explanation as to how atoms form covalent bonds is that one unpaired electron in the orbital of one atom overlaps with one in another atom. Sometimes atoms promote electrons in the same energy level to form more covalent bonds. For example, if an atom wants to make three covalent bonds but has a full 3s2 shell and a 3p1 shell, it can promote one of its 3s2 electrons so that an electron from the other atoms can fill the 3s shell and pair with the new 3p2 shell.
Sometimes promotion does not occur and that means different compounds can be made such as PCl3 or PCl5.

A lone pair of electrons is a pair of electrons from the same energy sub-level uninvolved in bonding. Sometimes these can form something called a coordinate bond, which contains a shared pair of electrons where both come from one atom. The lone pair of electrons is “donated” into the empty orbital of another atom to form a coordinate bond.

This is an example of a coordinate (sometimes called dative) bond between ammonia and a H+ ion which has an empty orbital. The lone pair on the ammonia overlaps with this H+ ion and donates its electrons. Both electrons come from the ammonia’s lone pair so it is a coordinate bond. This is demonstrated with an arrow. The diagram is missing an overall charge of + on the ammonium ion it produces. Coordinate bonds act the same as covalent bonds.
Once you have your covalent bonds, you need to know about covalent substances and their properties. There are two types of covalent substance: simple covalent (molecular) and macromolecular (giant covalent).
Molecular simply means that the formula for the compound or element describes exactly how many atoms are in one molecule, e.g. H2O. Molecular covalent crystalline substances usually exist as single molecules such as iodine or oxygen. They are usually gases or liquids at room temperature but can be low melting point solids.
Solid molecular covalent solids are crystalline so can be called molecular covalent crystals. Iodine and ice are examples of these. Iodine (shown below) has a regular arrangement which makes it a crystalline substance and water, as ice, has a crystalline structure as well.

The properties of these crystals are that they have low melting points, are very brittle due to the lack of strong bonds holding them together and also do not conduct electricity since no ions are present.
The other kind of covalent substance you need to know is macromolecular. This includes giant covalent structures such as diamond or graphite, which are allotropes of carbon. Non-metallic elements and compounds usually form these crystalline structures with a regular arrangement of atoms.
Allotropes are different forms of the same element in the same physical state.
Diamond is the hardest naturally occurring substance on earth therefore is good for cutting glass and drilling and mining. It has a high melting point due to the many covalent bonds which require a lot of energy to break. Each carbon has four of these bonds joining it to four others in a tetrahedral arrangement with a bond angle of 109.5 degrees and it does not conduct electricity or heat because there are no ions free to move.
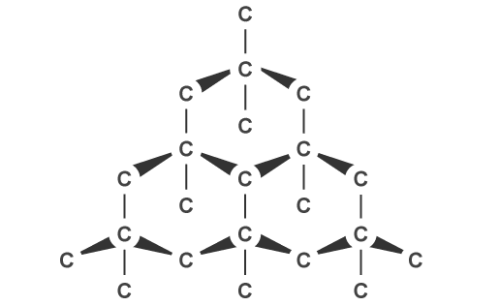
Graphite, on the other hand, can conduct electricity. This is because it has delocalised electrons between the layers which move and carry charge. Carbon atoms within the structure are only bonded to three others in a hexagonal arrangement with a bond angle of 120 degrees. Since only three of carbon’s unpaired electrons are used in bonding, the fourth becomes delocalised and moves between the layers of graphite causing weak attractions, explaining why it can conduct electricity.

Graphite’s layered structure and the weak forces of attractions between it make it a good lubricant and ideal for pencil lead because the layers can slide over each other. The attractions can be broken easily but the covalent bonds within the layers give graphite a high melting point due to the amount of energy needed to break them.
SUMMARY
Covalent bonding involves one or more shared pairs of electrons between two atoms. Covalent bonds mostly occur between non-metals but sometimes metals can form covalent bonds.
Single covalent bonds share just one pair of electrons. Double covalent bonds share two. Triple covalent bonds share three.
Each atom usually provides one electron – unpaired in the orbital – in the bond. The number of unpaired electrons in an atom usually shows how many bonds it can make but sometimes atoms promote electrons to fit in more. Covalent bonds are represented with lines between the atoms.
Dot and cross diagrams use dots and crosses to demonstrate that the electrons come from different places and often only the outer shell is shown.
The simple explanation as to how atoms form covalent bonds is that one unpaired electron in the orbital of one atom overlaps with one in another atom. Sometimes atoms promote electrons in the same energy level to form more covalent bonds.
Sometimes promotion does not occur and that means different compounds can be made such as PCl3 or PCl5.
A lone pair of electrons is a pair of electrons from the same energy sub-level uninvolved in bonding. Sometimes these can form something called a coordinate bond, which contains a shared pair of electrons where both come from one atom. The lone pair of electrons is “donated” into the empty orbital of another atom to form a coordinate bond.
The formation of ammonium is an example of this.
There are two types of covalent substance: simple covalent (molecular) and macromolecular (giant covalent).
Molecular simply means that the formula for the compound or element describes exactly how many atoms are in one molecule, e.g. H2O. Molecular covalent crystalline substances usually exist as single molecules such as iodine or oxygen. They are usually gases or liquids at room temperature but can be low melting point solids.
Solid molecular covalent solids are crystalline so can be called molecular covalent crystals. Iodine and ice are examples of these.
The properties of these crystals are that they have low melting points, are very brittle due to the lack of strong bonds holding them together and also do not conduct electricity since no ions are present.
Giant covalent structures such as diamond or graphite are allotropes of carbon. Allotropes are different forms of the same element in the same physical state.
Diamond has a high melting point due to the many covalent bonds which require a lot of energy to break. Each carbon has four of these bonds joining it to four others in a tetrahedral arrangement with a bond angle of 109.5 degrees and it does not conduct electricity or heat because there are no ions free to move.
Graphite can conduct electricity. This is because it has delocalised electrons between the layers which move and carry charge. Carbon atoms within the structure are only bonded to three others in a hexagonal arrangement with a bond angle of 120 degrees. Since only three of carbon’s unpaired electrons are used in bonding, the fourth becomes delocalised and moves between the layers of graphite causing weak attractions, explaining why it can conduct electricity.
Graphite’s layered structure and the weak forces of attractions between it make it a good lubricant and ideal for pencil lead because the layers can slide over each other. The attractions can be broken easily but the covalent bonds within the layers give graphite a high melting point due to the amount of energy needed to break them.
Happy studying!

My friend sent this to her Professor today
What comes to mind when you think of alcohol? Probably alcoholic drinks like beer or wine. But in organic chemistry alcohols are an important and versatile family of compounds. In this episode of Crash Course Organic Chemistry, we’ll use alcohols as a starting point to get other types of compounds like ethers, epoxides, and more!
-
 falcon1strike liked this · 3 years ago
falcon1strike liked this · 3 years ago -
 changoramos liked this · 3 years ago
changoramos liked this · 3 years ago -
 cstein1979 reblogged this · 3 years ago
cstein1979 reblogged this · 3 years ago -
 cstein1979 liked this · 3 years ago
cstein1979 liked this · 3 years ago -
 anactualamphibian reblogged this · 4 years ago
anactualamphibian reblogged this · 4 years ago -
 eternalodetoathena liked this · 4 years ago
eternalodetoathena liked this · 4 years ago -
 pleasurehunter2000 liked this · 4 years ago
pleasurehunter2000 liked this · 4 years ago -
 eternallyyours666 liked this · 4 years ago
eternallyyours666 liked this · 4 years ago -
 exactlymellowstudent liked this · 4 years ago
exactlymellowstudent liked this · 4 years ago -
 themajoriconsworld liked this · 4 years ago
themajoriconsworld liked this · 4 years ago -
 electricalxpert reblogged this · 4 years ago
electricalxpert reblogged this · 4 years ago -
 amateurchemstudent reblogged this · 4 years ago
amateurchemstudent reblogged this · 4 years ago -
 amateurchemstudent liked this · 4 years ago
amateurchemstudent liked this · 4 years ago -
 philameangrey reblogged this · 4 years ago
philameangrey reblogged this · 4 years ago -
 philameangrey liked this · 4 years ago
philameangrey liked this · 4 years ago -
 japanerinihongo liked this · 4 years ago
japanerinihongo liked this · 4 years ago



